.
Introduction
Owing to its involvement in numerous biochemical pathways and its actions in hearing, taste, cell mediated immunity, anti-oxidant activity, cell membrane stabilization, anti-inflammatory and anti-viral effects, zinc plays a pivotal role in disease pathogenesis (Gaby, 2011). Zinc is a structural element in enzymes, receptors, cytokines, growth factors and transcription factors that regulate cellular signalling and other biological activities (Fukada et al., 2011). Zinc deficiency symptoms are illustrated in Table 1.
Table 1. Zinc Deficiency Symptoms

(Prasad, 1985)
Long COVID symptoms include many of those illustrated in the above table. However, post-viral syndrome with many of the same sequelae has been a feature of many different viruses and has been described in recent as well as past literature for many years (Stefano, 2021) (Dani et al., 2021). The extended duration of long COVID symptoms may be potentially attributed in part to compromised cell and antibody mediated immune responses, as well as lymphopenia and thymic atrophy – characteristics common to zinc deficiency (Fukada et al., 2011).
One potential reason as to why long COVID symptoms persist could be that zinc and other nutritional deficiencies are not being addressed with conventional “standard of care” medicine. This is generally defined here as isolation, palliation, masking, social-distancing, over-the-counter antipyretics, analgesics or antitussives and fluid administration during initial viral symptoms. Then as symptoms increase in severity, hospitalization and administration of medications such as Remdesivir, Dexamethasone and other anti-virals (COVID-19 Treatment Guidelines).
Correcting zinc deficiency requires that zinc adequately penetrates into all cells. The co-administration of zinc ionophores, augments repletion and subsequent recovery, in addition to providing an auxiliary of therapeutic benefits (Celik, Gencay & Ocsoy, 2021). In Chinese medicine pathophysiology, viral infection is referred to as “wind-heat invasion” with long-term post-viral symptoms referred to as “latent or smoldering heat lingering in the interior” after improperly cleared “wind-heat” (Maciocia, 1994). Chinese herbs have been used as antivirals for many years to reduce severity and sequelae of viral infections and their use in a COVID context is due in part to their quercetin and other zinc ionophore content which can increase intracellular zinc levels (Luo et al., 2020), (Bensky & Gamble, 1993).
Methods
A search of the existing literature using the keywords: Long COVID, COVID-19, zinc, zinc ionophores, epigallocatechin galate, quercetin, hydroxychloroquine, ivermectin, clioquinol, pyrithione, curcumin, tannic acid, and luteolin was performed utilizing PubMED, Web of Science and PubChem internet databases. Articles for review were confined to meta-analyses, review articles and clinical trials discussing ionophoric activity of the aforementioned molecules, or mechanisms in which they work. Evidence and information regarding the functions and applications of each molecule was consolidated and presented, followed by our professional opinions on their potential use in research and practice. While this is only a general review, we feel it worth mentioning in order to stimulate further inquiry into their possible use as therapeutics in conjunction with existing protocols of treatment.
Polyphenolic Molecules with Strong Zinc Ionophoric Activity
Clioquinol
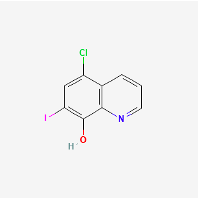
(PubChem Clioquinol: https://pubchem.ncbi.nlm.nih.gov/compound/Clioquinol)
Clioquinol (5-chloro-7-iodo-8-hydroxyquinoline) was used in the past as an oral anti-parasitic agent until the discovery that it induced mitochondrial and neurotoxicity in the presence of zinc when orally administered (Arbiser et al., 1998). While it can be used topically to treat prostate cancer (Costello & Franklin, 2019), it would be of dubious benefit in the short term treatment of long covid if given internally due to these toxicity concerns.
Pyrithione
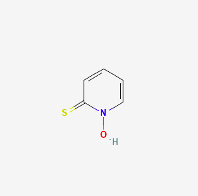
(PubChem Pyrithione: https://pubchem.ncbi.nlm.nih.gov/compound/Pyrithione)
Zinc Pyrithione finds use in medicine as a topical anti-fungal for seborrheic dermatitis (Barak-Shinar & Green, 2018). However, it is toxic via any other route of administration and therefore of no benefit for long COVID treatment (Scientific Committee on Consumer Safety, Opinion in Zinc Pyrithione, 2013).
Epigallocatechin Galate (EGCG)
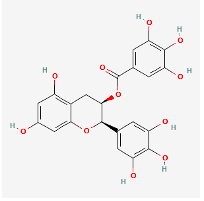
(PubChem Epigallocatechin Gallate: https://pubchem.ncbi.nlm.nih.gov/compound/Epigallocatechin-gallate)
Epigallocatechin Galate (EGCG) is a water soluble polyphenolic catechin found most abundantly in green tea that exhibits strong zinc ionophoric activity (Dabbagh-Bazarbachi et al., 2014). In cancer treatment and prevention it has been shown to down-regulate both COX-2 and NFkB, lower inflammation, and up-regulate TNFa, which increases cancer cell apoptosis (Ramos, 2008). EGCG has also been shown to have potent antiviral activity to a number of viruses including HCV, HIV-1, influenza, zika and others via viral binding interference, inhibition of reverse transcriptase and other mechanisms (Steinmann et al., 2013; Mhatre et al., 2021). When combined with theaflavin (an active ingredient of black tea), there is a dose-dependent inhibition of SARS-CoV-2 3CL-protease, an enzyme critical to viral replication (Jang et al., 2020). During viral infection with H5N1, early administration of EGCG has been demonstrated to inhibit membrane fusion and viral entry into cells, subsequently lowering the viral titre (Saadh & Aldalaen, 2021). Coadministration of EGCG with zinc strengthens this inhibition (Saadh & Aldalaen, 2021). A common theme to long COVID syndrome is the chronic inflammation that remains in the wake of the initial infection. The inflammatory mechanism of long COVID is comparable to that of rheumatoid arthritis in that the increased amounts of inflammatory cytokine IL-1b cause down-stream increases in IL-6 and IL-8 which add to the overall inflammatory burden (Singh et al., 2016). When deployed early or prophylactically, EGCG may help to curb the length of time that this inflammation persists (Chourasia et al., 2021).
Quercetin
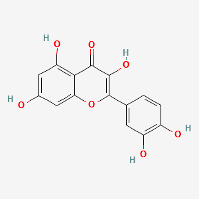
(PubChem Quercetin: https://pubchem.ncbi.nlm.nih.gov/compound/Quercetin)
Quercetin (QCT) is a water insoluble bioflavonoid that is found in many fruits, vegetables and herbs, with concentrations in typical foods as listed in Table 2.
Table 2. Common Sources of Quercetin
(Cione et al., 2019)
Quercetin has desirable therapeutic characteristics which include immunomodulation, anti-inflammatory and antiviral capabilities, thrombin-inhibitory actions and strong antioxidant capacity (Di Pierro et al., 2021). Many of the Chinese medicinal herbs that are currently being employed as part of multimodal treatments for COVID-19 in China also contain quercetin in beneficial amounts (Luo et al., 2020), (Bensky & Gamble, 1993). When quercetin is combined with very large doses of vitamin C, its anti-inflammatory effects are increased, and with zinc added, the immunomodulatory effect may be strengthened via inhibition of RNA dependant RNA polymerase, which prevents covid viral replication (Pal et al., 2021). In addition to being well tolerated and lacking in any side effects, quercetin has also been shown to help ameliorate chronic fatigue and poor appetite, which may help to improve long COVID outcomes (Di Pierro et al., 2021).
Curcumin
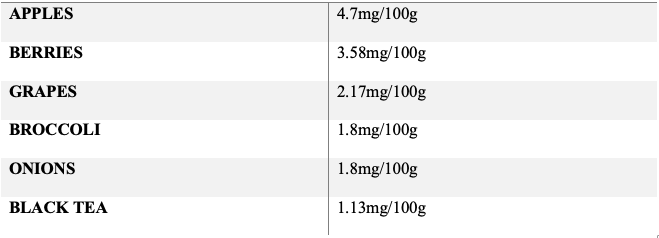
(PubChem Curcumin: https://pubchem.ncbi.nlm.nih.gov/compound/Curcumin)
Curcumin is a phenolic compound coming from turmeric (curcuma longa) and part of the ginger family of plants (Zingiberaceae) that has widespread use in East Asia as a food ingredient, additive, Chinese medicinal (Jiang Huang – blood invigorator), Ayurvedic medicinal and anti-inflammatory (Cione et al., 2019; Bensky & Gamble, 1993). Curcumin exhibits antiviral activity against enveloped viruses such as dengue and hepatitis C by interfering with the binding of viruses to cell receptors (Keflie & Biesalski, 2021). However, due to its low solubility, other methods of increasing its bioavailability should be explored. Many polyphenols, including curcumin, form chelates with various transition metals such as zinc cations (Celik, Gencay & Ocsoy, 2021). This curcumin-zinc (II) complex may increase the bioavailability and effectiveness of zinc (Sareen, Jain & Dhar, 2016). There have not been any studies examining curcumin as a zinc ionophore in the context of SARS-CoV2 infection; thus zinc and curcumin co-supplementation may be of interest in future trials.
Tannic Acid
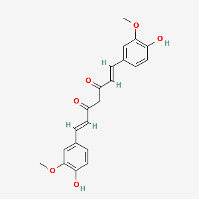
(PubChem Tannic Acid: https://pubchem.ncbi.nlm.nih.gov/compound/Tannic-acid)
Tannic acid (or Chinese tannin) is one of the polyphenols in tea which has similar zinc ionophoric actions. Similar to quercetin and EGCG, tannic acid also binds and inactivates the viral cysteine proteases PLpro (papain-like protease) and 3CLpro (chymotrypsin-like protease), which blocks viral replication in the host cell cytosol (Kaul et al., 2021). In addition, tannic acid and other polyphenols exhibit broad spectrum antibacterial and antifungal characteristics (Chung et al, 1998). These anti-microbial functions can help to prevent bacterial superinfection in at-risk patients suffering from long COVID.
Luteolin
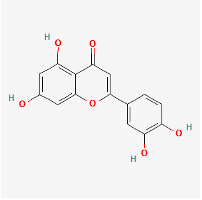
(PubChem Luteolin: https://pubchem.ncbi.nlm.nih.gov/compound/Luteolin)
Luteolin is a bioactive flavone present in many medicinal herbs, common fruits and vegetables such as onions, cabbage, peppers, apple skins, lemongrass, dandelion, perilla leaf and chrysanthemum (Aziz, Kim & Cho, 2018). Its therapeutic properties include anti-inflammatory, antimicrobial, antioxidant and neuroprotective functions, and it is non-toxic (Aziz, Kim & Cho, 2018). As with other bioflavonoids like quercetin, luteolin may inhibit a variety of inflammatory cytokines including interleukins (IL) 1b,2,4,5,6,8,12,13,17 and TNF-a, inhibit COX-2 enzyme, as well as stimulate the release of IL10, an anti-inflammatory cytokine, and inhibit histamine release from mast cells (Seelinger, Merfort & Schempp, 2008). There is speculation that long COVID symptoms could be related to abnormal hyperactivation of dysfunctional mast cells (Afrin, Weinstock & Molderings, 2020), and thus downregulation of mast cell activation or mast cell mediated inflammation may aid with long COVID symptomatology.
Hydroxychloroquine and Ivermectin
(PubChem Hydroxychloroquine: https://pubchem.ncbi.nlm.nih.gov/compound/Hydroxychloroquine)
(PubChem Ivermectin: https://pubchem.ncbi.nlm.nih.gov/compound/Ivermectin)
Both hydroxychloroquine and ivermectin have been at the center of controversy surrounding SARS-CoV-2 treatment since nearly the beginning of the pandemic. While it is beyond the scope of this paper to comment on the possible reasons for this, it is important to mention that both drugs originated from natural sources, and display zinc ionophoric activity (Butters & Whitehouse, 2021). Although previously mentioned ionophores display affinities for other transition metals such as iron and copper to some degree as well, hydroxychloroquine appears to be specific to zinc which may be advantageous as zinc deficiency is of the utmost concern in the context of both acute SARS-CoV-2 and subsequent long COVID treatment (Xue et al., 2014).
Ivermectin was first discovered by professor Satoshi Omura in 1975 from a soil sample containing fermentation products from Streptomyces avermitilis bacteria. Due to its versatility and benefit in treating parasitosis, viruses and some cancers, Dr. Omura was awarded the 2015 Nobel Prize in physiology or medicine for this discovery (Crump, 2017). While ivermectin was initially used to treat onchocerciasis and strongyloidiasis, it has found off label uses in treating cancer, asthma and viral infections from dengue, encephalitis, HIV and several RNA viruses (including SARS-CoV-2) by the blockade of nuclear trafficking of viral proteins (Crump, 2017). It has also been postulated to be a zinc ionophore via two ivermectin molecules reacting in a head to tail orientation which would aid in escorting zinc ions into the cell cytoplasm (Zaidi & Dehgani-Mobaraki, 2022). Being a zinc ionophore would account for one of its many reasons for inclusion into doctor led clinical protocols and trials such as the Front Line Covid Critical Care Alliance (FLCCC) and the Canadian Covid Care Alliance (CCCA) (Rizzo, 2020).
Discussion
Molecules that have ionophoric characteristics are usually described as having a hydrophilic pocket that allow for one or more binding sites for cations, yet the outer surface of the molecule is hydrophobic. This allows a complex to form around the ions, making them able to transport across cell membranes, which alters electrolyte balance (Freedman, 2012). The rationale for concentrating on zinc and its repletion in this paper is due to both the symptoms of long COVID converging with many of the symptoms of zinc deficiency as illustrated in Table 1, as well as widespread zinc deficiency in many global elderly and vulnerable populations (Mocchegiani et al., 2013). There are various causes of zinc deficiency which include genetics, nutrition, iatrogenic factors and chronic diseases such as ulcerative colitis and alcoholism (Saper & Rash, 2009). However, the most effective method of treatment for long COVID in our opinion is preventative/ prophylactic in nature or one which reduces the symptom severity should COVID-19 infection occur.
Although zinc status is usually checked via plasma zinc values (morning fasting lower limit: 10.7mmol/L), plasma does not indicate cellular zinc concentrations (Maret & Sandstead, 2006). The total body pool of zinc is between 2-3g in adults, yet this pool is not uniformly distributed across all tissues. Those tissues with high rates of cellular turnover, such as the liver and blood cells (granulocytes, lymphocytes and platelets) are likely more sensitive to mild or moderate deficiencies in zinc. For example, it has been demonstrated that elderly subjects can have normal plasma zinc levels, yet low leukocyte zinc which reflects immune impairment (low thymulin activity) (Prasad et al., 1988).
The antiviral mechanisms of zinc appear to vary with virus type. For example, intracellular Zn2+ ions have been shown to inhibit SARS CoV-1 RNA dependent RNA Polymerase (RdRp) template binding and elongation (Read et al., 2019), whereas free zinc binds to HSV virions, preventing infection by inhibition of viral membrane fusion (Arens & Travis, 2000).
Although zinc deficiency is far more common globally than its excess (Pal et al., 2021), a word about potential zinc toxicity with over-supplementation is also required as it can lead to immunosuppression (Chandra, 1984). Excessive zinc supplementation can induce copper deficiency, which can impair antibody production by T-lymphocytes (Blakley & Hamilton, 1987). However, the induction of copper deficiency due to excessive zinc supplementation is an issue that most nutritionally minded clinicians are aware of, and mitigate via concurrent copper supplementation and zinc level monitoring.
Zinc supplementation on its own takes time to be effective due to slow fractional absorption (Maares & Haase, 2020), highlighting the need for co-supplementation with zinc ionophores.
In theory, if total body cellular zinc levels can be brought up quickly during or soon after acute SARS COV-2 infection, there may be reduced risk of developing long COVID or the symptom severity and disease progression may be lessened. The two readily available, over-the-counter and non-toxic bioflavonoids quercetin (QCT) and epigallocatechin galate (EGCG), are both strong zinc ionophores (Dabbagh-Bazarbachi et al., 2014). Both are found in abundance in the Japanese diet (QCT in vegetables and EGCG in green tea) in addition to zinc rich foods such as oysters. This may help to explain why, despite the population density of Japan, the lower case loads of COVID-19 are being observed. Both quercetin and zinc are a part of doctor led recommended treatment protocols that have been developed and adopted by organizations such as the Front Line COVID-19 Critical Care Alliance (FLCCC) and Canadian COVID Care Alliance (CCCA) as part of multi-drug/ nutrient treatment and preventative regimens such as I-MASK+, MATH+ and I-RECOVER protocols which also include key nutrients such as vitamin C, vitamin D and thiamine . I-RECOVER protocol in particular is being used for long COVID.
Conclusion
While current measures such as vaccination, social distancing, masking and lockdowns are a part of the solution to the global pandemic, none of them will enhance nutrient repletion needed to reduce susceptibility to, or symptomatology of, COVID-19. In China, the pharmacological properties of combined Chinese herbal prescriptions are well recognized and are helping to treat COVID-19. Likewise, poly-modal approaches to this complex pandemic that include the use of zinc and its ionophores must also be employed in order to help reduce the overall burden to global society that this pandemic has created.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflicts of interest.
References
Afrin, L. B., Weinstock, L. B., & Molderings, G. J. (2020). Covid-19 hyperinflammation and post-Covid-19 illness may be rooted in mast cell activation syndrome. International journal of infectious diseases : IJID : official publication of the International Society for Infectious Diseases, 100, 327–332. https://doi.org/10.1016/j.ijid.2020.09.016
Arbiser, J. L., Kraeft, S. K., van Leeuwen, R., Hurwitz, S. J., Selig, M., Dickersin, G. R., Flint, A., Byers, H. R., & Chen, L. B. (1998). Clioquinol-zinc chelate: a candidate causative agent of subacute myelo-optic neuropathy. Molecular medicine (Cambridge, Mass.), 4(10), 665–670.
Arens, M., & Travis, S. (2000). Zinc salts inactivate clinical isolates of herpes simplex virus in vitro. Journal of clinical microbiology, 38(5), 1758–1762. https://doi.org/10.1128/JCM.38.5.1758-1762.2000
Aziz, N., Kim, M. Y., & Cho, J. Y. (2018). Anti-inflammatory effects of luteolin: A review of in vitro, in vivo, and in silico studies. Journal of ethnopharmacology, 225, 342–358. https://doi.org/10.1016/j.jep.2018.05.019
Barak-Shinar, D., & Green, L. J. (2018). Scalp Seborrheic Dermatitis and Dandruff Therapy Using a Herbal and Zinc Pyrithione-based Therapy of Shampoo and Scalp Lotion. The Journal of clinical and aesthetic dermatology, 11(1), 26–31.
Bensky D., Gamble A., (1993), Chinese Herbal Medicine Materia Medica (Revised Edition), Seattle, WA, Eastland Press Inc.
Blakley, B. R., & Hamilton, D. L. (1987). The effect of copper deficiency on the immune response in mice. Drug-nutrient interactions, 5(2), 103–111.
Butters, D., & Whitehouse, M. (2021). COVID-19 and nutriceutical therapies, especially using zinc to supplement antimicrobials. Inflammopharmacology, 29(1), 101–105. https://doi.org/10.1007/s10787-020-00774-8
Celik, C., Gencay, A., & Ocsoy, I. (2021). Can food and food supplements be deployed in the fight against the COVID 19 pandemic?. Biochimica et biophysica acta. General subjects, 1865(2), 129801. https://doi.org/10.1016/j.bbagen.2020.129801
Chandra R. K. (1984). Excessive intake of zinc impairs immune responses. JAMA, 252(11), 1443–1446.
Chourasia, M., Koppula, P. R., Battu, A., Ouseph, M. M., & Singh, A. K. (2021). EGCG, a Green Tea Catechin, as a Potential Therapeutic Agent for Symptomatic and Asymptomatic SARS-CoV-2 Infection. Molecules (Basel, Switzerland), 26(5), 1200. https://doi.org/10.3390/molecules26051200
Chung, K. T., Wong, T. Y., Wei, C. I., Huang, Y. W., & Lin, Y. (1998). Tannins and human health: a review. Critical reviews in food science and nutrition, 38(6), 421–464. https://doi.org/10.1080/10408699891274273
Cione, E., La Torre, C., Cannataro, R., Caroleo, M. C., Plastina, P., & Gallelli, L. (2019). Quercetin, Epigallocatechin Gallate, Curcumin, and Resveratrol: From Dietary Sources to Human MicroRNA Modulation. Molecules (Basel, Switzerland), 25(1), 63. https://doi.org/10.3390/molecules25010063
Costello, L. C., & Franklin, R. B. (2019). A Proposed Efficacious Treatment with Clioquinol (Zinc Ionophore) and Cabergoline (Prolactin Dopamine Agonist) for the Treatment of Terminal Androgen-independent Prostate Cancer. Why and How?. Journal of clinical research in oncology, 2(1), https://asclepiusopen.com/journal-of-clinical-research-in-oncology/volume-2-issue-1/1.pdf.
COVID-19 Treatment Guidelines Panel. Coronavirus Disease 2019 (COVID-19) Treatment Guidelines. National Institutes of Health. Available at https://www.covid19treatmentguidelines.nih.gov/. Accessed (April 9, 2022).
Crump A. (2017). Ivermectin: enigmatic multifaceted ‘wonder’ drug continues to surprise and exceed expectations. The Journal of antibiotics, 70(5), 495–505. https://doi.org/10.1038/ja.2017.11
Dabbagh-Bazarbachi, H., Clergeaud, G., Quesada, I. M., Ortiz, M., O’Sullivan, C. K., & Fernández-Larrea, J. B. (2014). Zinc ionophore activity of quercetin and epigallocatechin-gallate: from Hepa 1-6 cells to a liposome model. Journal of agricultural and food chemistry, 62(32), 8085–8093. https://doi.org/10.1021/jf5014633
Dani, M., Dirksen, A., Taraborrelli, P., Torocastro, M., Panagopoulos, D., Sutton, R., & Lim, P. B. (2021). Autonomic dysfunction in ‘long COVID’: rationale, physiology and management strategies. Clinical medicine (London, England), 21(1), e63–e67. https://doi.org/10.7861/clinmed.2020-0896
Di Pierro, F., Derosa, G., Maffioli, P., Bertuccioli, A., Togni, S., Riva, A., Allegrini, P., Khan, A., Khan, S., Khan, B. A., Altaf, N., Zahid, M., Ujjan, I. D., Nigar, R., Khushk, M. I., Phulpoto, M., Lail, A., Devrajani, B. R., & Ahmed, S. (2021). Possible Therapeutic Effects of Adjuvant Quercetin Supplementation Against Early-Stage COVID-19 Infection: A Prospective, Randomized, Controlled, and Open-Label Study. International journal of general medicine, 14, 2359–2366. https://doi.org/10.2147/IJGM.S318720
Freedman JC (2012) Chapter 4 – Ionophores in planar lipid bilayers (pp 61–66). In: Sperelakis N (ed) Cell Physiology Source Book, 4th edn. Academic Press.
Fukada, T., Yamasaki, S., Nishida, K., Murakami, M., & Hirano, T. (2011). Zinc homeostasis and signaling in health and diseases: Zinc signaling. Journal of biological inorganic chemistry : JBIC : a publication of the Society of Biological Inorganic Chemistry, 16(7), 1123–1134. https://doi.org/10.1007/s00775-011-0797-4
Gaby, A. R. (2011). Chapter 31., Zinc. In Nutritional Medicine (1st ed., pp. 151–158) Concord, NH: Fritz Perlberg Publishing.
Heinz, S. A., Henson, D. A., Austin, M. D., Jin, F., & Nieman, D. C. (2010). Quercetin supplementation and upper respiratory tract infection: A randomized community clinical trial. Pharmacological research, 62(3), 237–242. https://doi.org/10.1016/j.phrs.2010.05.001
Jang, M., Park, Y. I., Cha, Y. E., Park, R., Namkoong, S., Lee, J. I., & Park, J. (2020). Tea Polyphenols EGCG and Theaflavin Inhibit the Activity of SARS-CoV-2 3CL-Protease In Vitro. Evidence-based complementary and alternative medicine : eCAM, 2020, 5630838. https://doi.org/10.1155/2020/5630838
Kaul, R., Paul, P., Kumar, S., Büsselberg, D., Dwivedi, V. D., & Chaari, A. (2021). Promising Antiviral Activities of Natural Flavonoids against SARS-CoV-2 Targets: Systematic Review. International journal of molecular sciences, 22(20), 11069. https://doi.org/10.3390/ijms222011069
Keflie, T. S., & Biesalski, H. K. (2021). Micronutrients and bioactive substances: Their potential roles in combating COVID-19. Nutrition (Burbank, Los Angeles County, Calif.), 84, 111103. https://doi.org/10.1016/j.nut.2020.111103
Luo, E., Zhang, D., Luo, H., Liu, B., Zhao, K., Zhao, Y., Bian, Y., & Wang, Y. (2020). Treatment efficacy analysis of traditional Chinese medicine for novel coronavirus pneumonia (COVID-19): an empirical study from Wuhan, Hubei Province, China. Chinese medicine, 15, 34. https://doi.org/10.1186/s13020-020-00317-x
Maares, M., & Haase, H. (2020). A Guide to Human Zinc Absorption: General Overview and Recent Advances of In Vitro Intestinal Models. Nutrients, 12(3), 762. https://doi.org/10.3390/nu12030762
Maciocia, G. (1994). Ch. 25. Myalgic encephalomyelitis. In The Practice of Chinese Medicine (1st ed., pp. 631- 655) Churchill Livingstone Publishing, UK.
Maret, W., & Sandstead, H. H. (2006). Zinc requirements and the risks and benefits of zinc supplementation. Journal of trace elements in medicine and biology : organ of the Society for Minerals and Trace Elements (GMS), 20(1), 3–18. https://doi.org/10.1016/j.jtemb.2006.01.006
Mhatre, S., Srivastava, T., Naik, S., & Patravale, V. (2021). Antiviral activity of green tea and black tea polyphenols in prophylaxis and treatment of COVID-19: A review. Phytomedicine : international journal of phytotherapy and phytopharmacology, 85, 153286. https://doi.org/10.1016/j.phymed.2020.153286
Mocchegiani, E., Romeo, J., Malavolta, M., Costarelli, L., Giacconi, R., Diaz, L. E., & Marcos, A. (2013). Zinc: dietary intake and impact of supplementation on immune function in elderly. Age (Dordrecht, Netherlands), 35(3), 839–860. https://doi.org/10.1007/s11357-011-9377-3
Pal, A., Squitti, R., Picozza, M., Pawar, A., Rongioletti, M., Dutta, A. K., Sahoo, S., Goswami, K., Sharma, P., & Prasad, R. (2021). Zinc and COVID-19: Basis of Current Clinical Trials. Biological trace element research, 199(8), 2882–2892. https://doi.org/10.1007/s12011-020-02437-9
Prasad A. S. (1985). Clinical and biochemical manifestations of zinc deficiency in human subjects. Journal of the American College of Nutrition, 4(1), 65–72. https://doi.org/10.1080/07315724.1985.10720067
PubChem [Internet]. Bethesda (MD): National Library of Medicine (US), National Center for Biotechnology Information; 2004-. PubChem Compound Summary for CID 2788, Clioquinol; [cited 2022 Jan. 6]. Available from: https://pubchem.ncbi.nlm.nih.gov/compound/Clioquinol
PubChem [Internet]. Bethesda (MD): National Library of Medicine (US), National Center for Biotechnology Information; 2004-. PubChem Compound Summary for CID 1570, Pyrithione; [cited 2022 Jan. 6]. Available from: https://pubchem.ncbi.nlm.nih.gov/compound/Pyrithione
PubChem [Internet]. Bethesda (MD): National Library of Medicine (US), National Center for Biotechnology Information; 2004-. PubChem Compound Summary for CID 65064, Epigallocatechin gallate; [cited 2022 Jan. 6]. Available from: https://pubchem.ncbi.nlm.nih.gov/compound/Epigallocatechin-gallate
PubChem [Internet]. Bethesda (MD): National Library of Medicine (US), National Center for Biotechnology Information; 2004-. PubChem Compound Summary for CID 5280343, Quercetin; [cited 2022 Jan. 6]. Available from: https://pubchem.ncbi.nlm.nih.gov/compound/Quercetin
PubChem [Internet]. Bethesda (MD): National Library of Medicine (US), National Center for Biotechnology Information; 2004-. PubChem Compound Summary for CID 969516, Curcumin; [cited 2022 Jan. 6]. Available from: https://pubchem.ncbi.nlm.nih.gov/compound/Curcumin
PubChem [Internet]. Bethesda (MD): National Library of Medicine (US), National Center for Biotechnology Information; 2004-. PubChem Compound Summary for CID 16129778, Tannic acid; [cited 2022 Jan. 6]. Available from: https://pubchem.ncbi.nlm.nih.gov/compound/Tannic-acid
PubChem [Internet]. Bethesda (MD): National Library of Medicine (US), National Center for Biotechnology Information; 2004-. PubChem Compound Summary for CID 5280445, Luteolin; [cited 2022 Jan. 6]. Available from: https://pubchem.ncbi.nlm.nih.gov/compound/Luteolin
PubChem [Internet]. Bethesda (MD): National Library of Medicine (US), National Center for Biotechnology Information; 2004-. PubChem Compound Summary for CID 3652, Hydroxychloroquine; [cited 2022 Jan. 6]. Available from: https://pubchem.ncbi.nlm.nih.gov/compound/Hydroxychloroquine
PubChem [Internet]. Bethesda (MD): National Library of Medicine (US), National Center for Biotechnology Information; 2004-. PubChem Compound Summary for CID 6321424, Ivermectin; [cited 2022 Jan. 6]. Available from: https://pubchem.ncbi.nlm.nih.gov/compound/Ivermectin
Ramos S. (2008). Cancer chemoprevention and chemotherapy: dietary polyphenols and signalling pathways. Molecular nutrition & food research, 52(5), 507–526. https://doi.org/10.1002/mnfr.200700326
Read, S. A., Obeid, S., Ahlenstiel, C., & Ahlenstiel, G. (2019). The Role of Zinc in Antiviral Immunity. Advances in nutrition (Bethesda, Md.), 10(4), 696–710. https://doi.org/10.1093/advances/nmz013
Rizzo E. (2020). Ivermectin, antiviral properties and COVID-19: a possible new mechanism of action. Naunyn-Schmiedeberg’s archives of pharmacology, 393(7), 1153–1156. https://doi.org/10.1007/s00210-020-01902-5
Saadh, M. J., & Aldalaen, S. M. (2021). Inhibitory effects of epigallocatechin gallate (EGCG) combined with zinc sulfate and silver nanoparticles on avian influenza A virus subtype H5N1. European review for medical and pharmacological sciences, 25(6), 2630–2636. https://doi.org/10.26355/eurrev_202103_25427
Saper, R. B., & Rash, R. (2009). Zinc: an essential micronutrient. American family physician, 79(9), 768–772.
Sareen, R., Jain, N., & Dhar, K. L. (2016). Curcumin-Zn(II) complex for enhanced solubility and stability: an approach for improved delivery and pharmacodynamic effects. Pharmaceutical development and technology, 21(5), 630–635. https://doi.org/10.3109/10837450.2015.1041042
Scientific Committee on Consumer Safety Opinion on Zinc Pyrithione, 18 June 2013. [(accessed on 8 February 2021)]. Available online: https://ec.europa.eu/health/scientific_committees/consumer_safety_en.
Seelinger, G., Merfort, I., & Schempp, C. M. (2008). Anti-oxidant, anti-inflammatory and anti-allergic activities of luteolin. Planta medica, 74(14), 1667–1677. https://doi.org/10.1055/s-0028-1088314
Singh, A. K., Umar, S., Riegsecker, S., Chourasia, M., & Ahmed, S. (2016). Regulation of Transforming Growth Factor β-Activated Kinase Activation by Epigallocatechin-3-Gallate in Rheumatoid Arthritis Synovial Fibroblasts: Suppression of K(63) -Linked Autoubiquitination of Tumor Necrosis Factor Receptor-Associated Factor 6. Arthritis & rheumatology (Hoboken, N.J.), 68(2), 347–358. https://doi.org/10.1002/art.39447
Stefano G. B. (2021). Historical Insight into Infections and Disorders Associated with Neurological and Psychiatric Sequelae Similar to Long COVID. Medical science monitor : international medical journal of experimental and clinical research, 27, e931447. https://doi.org/10.12659/MSM.931447
Steinmann, J., Buer, J., Pietschmann, T., & Steinmann, E. (2013). Anti-infective properties of epigallocatechin-3-gallate (EGCG), a component of green tea. British journal of pharmacology, 168(5), 1059–1073. https://doi.org/10.1111/bph.12009
Xue, J., Moyer, A., Peng, B., Wu, J., Hannafon, B. N., & Ding, W. Q. (2014). Chloroquine is a zinc ionophore. PloS one, 9(10), e109180. https://doi.org/10.1371/journal.pone.0109180
Zaidi, A. K., & Dehgani-Mobaraki, P. (2022). RETRACTED AND REPUBLISHED ARTICLE: The mechanisms of action of Ivermectin against SARS-CoV-2: An evidence-based clinical review article. The Journal of antibiotics, 75(2), 122. https://doi.org/10.1038/s41429-021-00430-5


